Compression Factor Exam Problem using Molar Volumes - Fully Explained!


Solved 1. What is compression factor? (2 marks) 2. Derive a

Solved A gas at 350 K and 12 atm has a molar volume 12 per
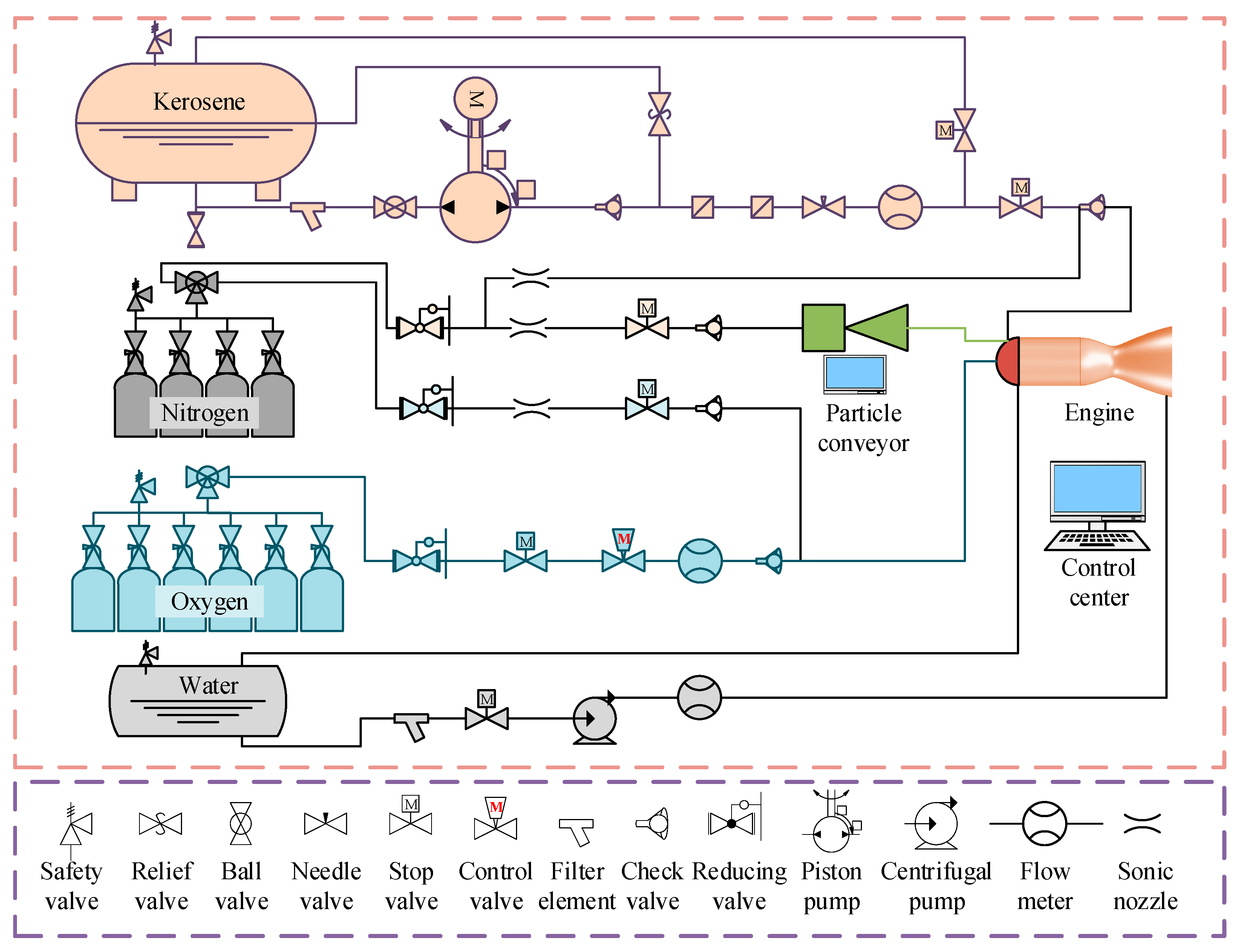
Aerospace, Free Full-Text

Energies, Free Full-Text

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

Molar Volume Calculated Two Different Ways

Is there a set of conditions at which the compression factor
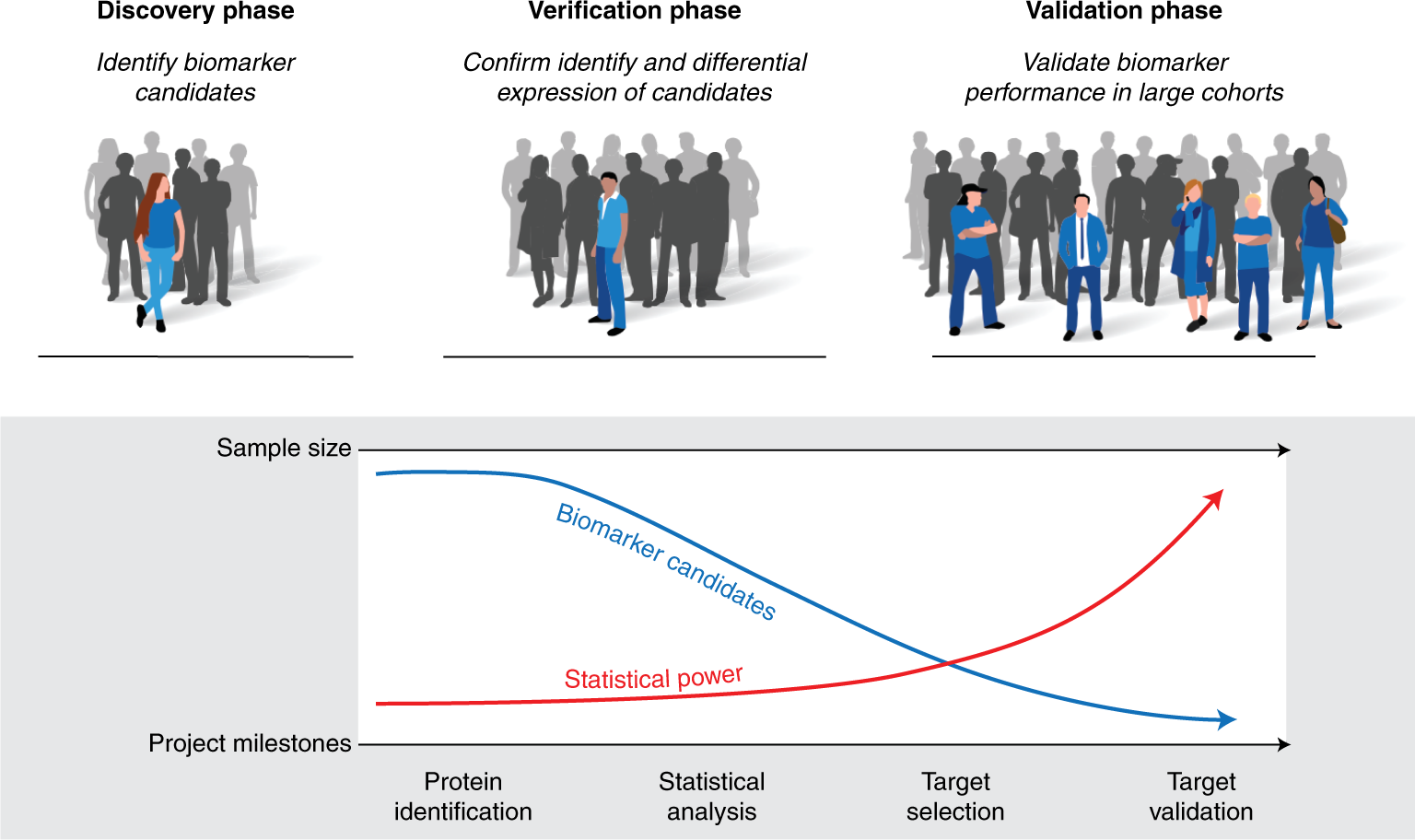
Tutorial: best practices and considerations for mass-spectrometry-based protein biomarker discovery and validation

The Ideal Gas Law

Physical Chemistry The Compression Factor (Z) [w/1 example]
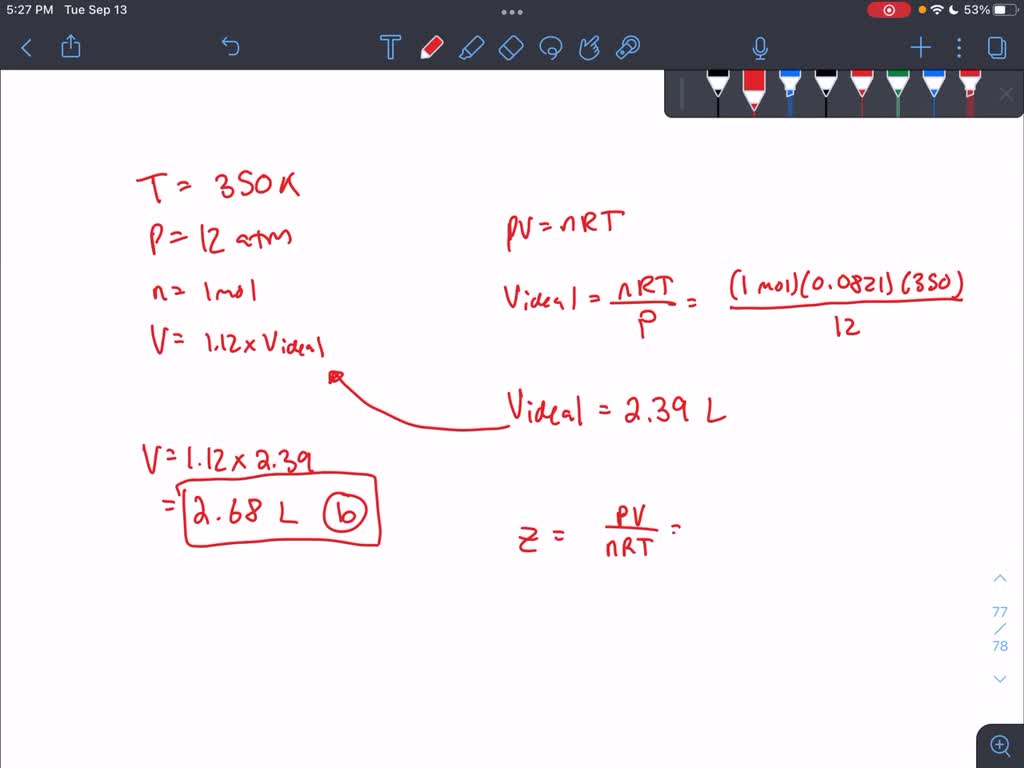
SOLVED: A gas at 350 K and 12 atm has a molar volume 12 per cent larger than that calculated from the perfect gas law. Calculate (a) The compression factor under these

Sizes of soil specimens for unconfined compression, direct shear and

Van der Waals Equation, Definition & Examples - Lesson

1.7: Connecting the van der Waals and the viral equations: the Boyle temperature - Chemistry LibreTexts

Compression Factor Exam Problem using Molar Volumes - Fully Explained!