The compressibility factor a real gas high pressure is:-1 - frac{Pb} {RT}1 + frac {RT} {Pb}11 + frac {Pb} {RT}

Click here:point_up_2:to get an answer to your question :writing_hand:the compressibility factor for a real gas at high pressure is
Click here👆to get an answer to your question ✍️ The compressibility factor a real gas high pressure is-1 - frac-Pb- -RT-1 - frac -RT- -Pb-11 - frac -Pb- -RT
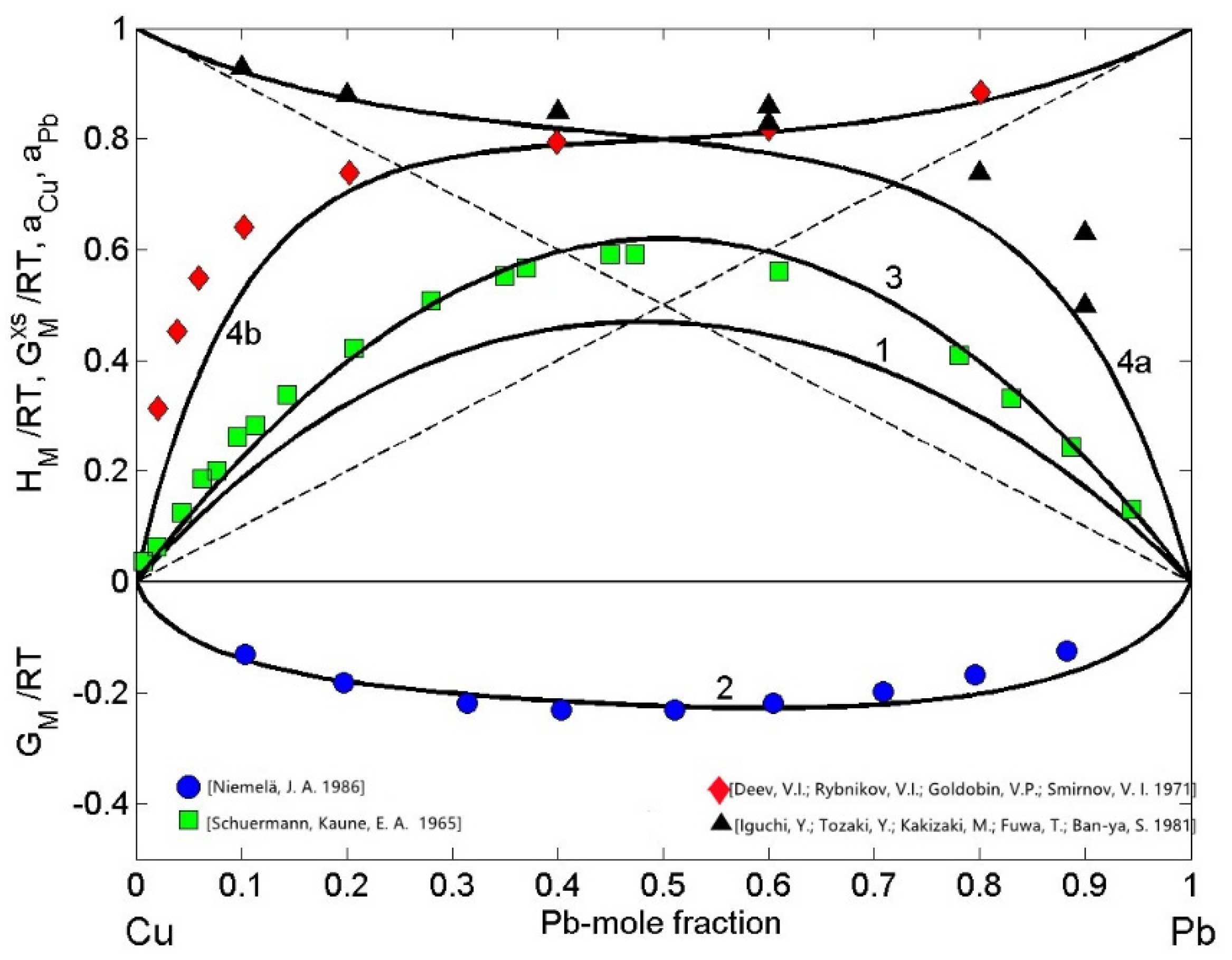
Metals, Free Full-Text
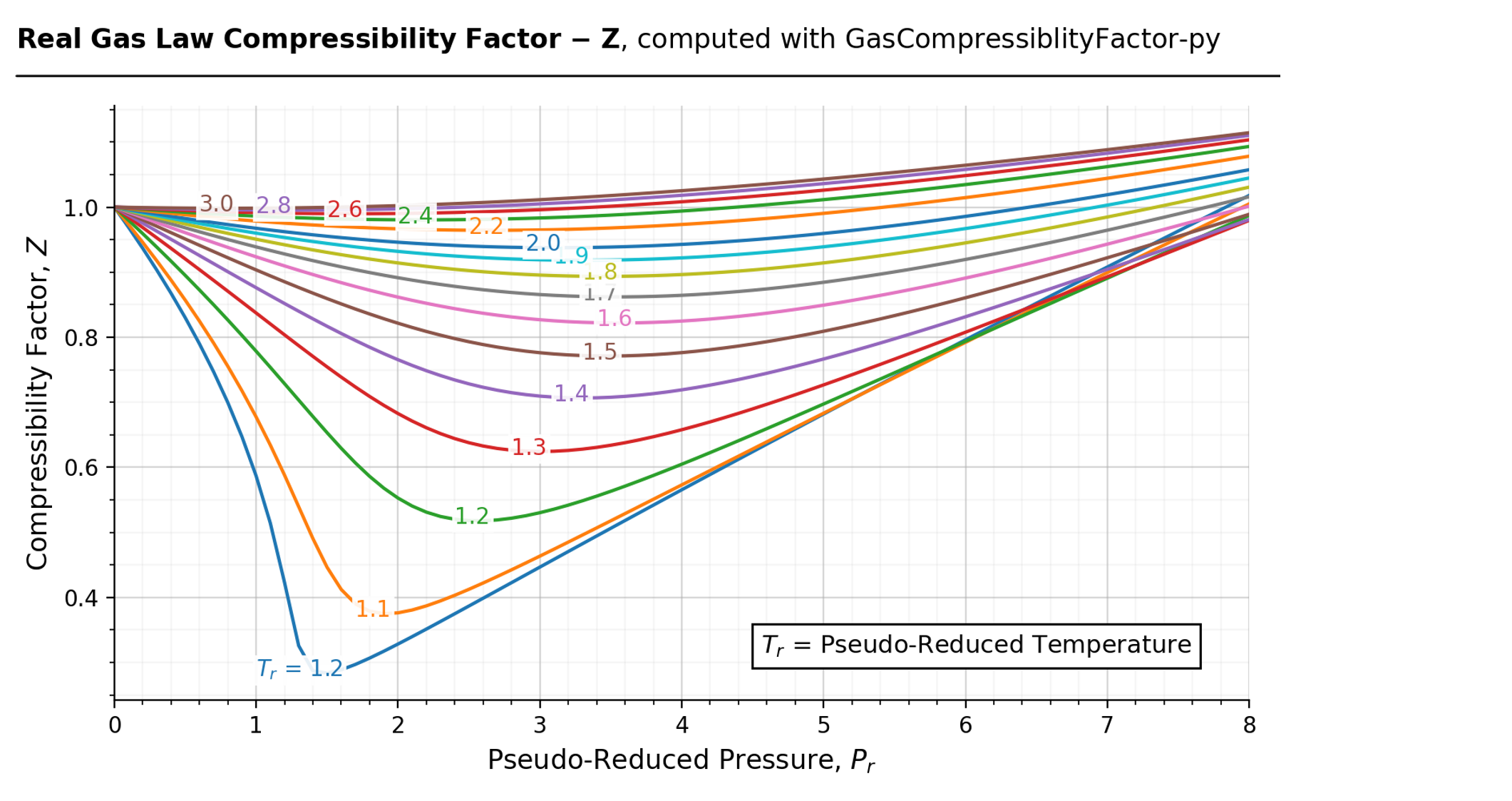
gascompressibility · PyPI
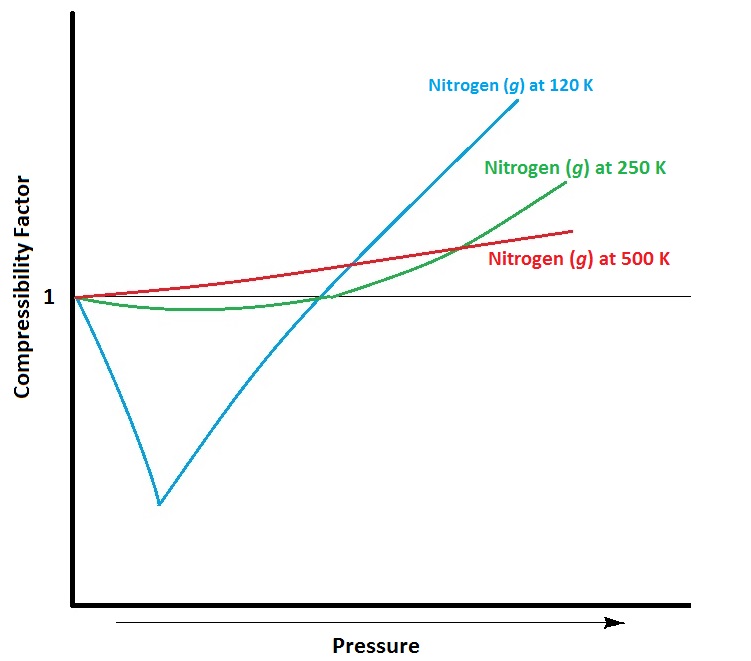
Real Gases – Introductory Chemistry – 1st Canadian Edition

Van der Waals Equation - Derivation, Relation Between Ideal Gas

Behavior of Gases

The compressibility factor for a real gas at high pressure is .

Deviation Of Real Gas From Ideal Gas Behavior

SOLVED: The compressibility factor for a real gas at high pressure is: A. 1 + RTpb B. 1 C. 1 + pbRT D. 1 - pbRT

The graph of compressibility factor Z vs P for one mole of a real

Thermodynamics of Physical and Chemical Transformations

The compressibility factor a real gas high pressure is:-1 - frac{Pb} {RT}1 + frac {RT} {Pb}11 + frac {Pb} {RT}

At high pressure, van der Waals' equation becomes

3.3: Real gas and compressibility factor - Engineering LibreTexts

P k nag solution by Shaikh Mohd Aslam - Issuu

Simple Equation Real Gas Compressibility Factor Z