The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is

Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor compressibility factor for onemole of a van der waals gas at 0c
Click here👆to get an answer to your question ✍️ The compression factor -compressibility factor- one mole of a van der Waals gas 0-C and 100 atm pressure is found to be 0-5- Assuming that the volume of a gas molecule is negligible- calculate the van der Waals- constant a
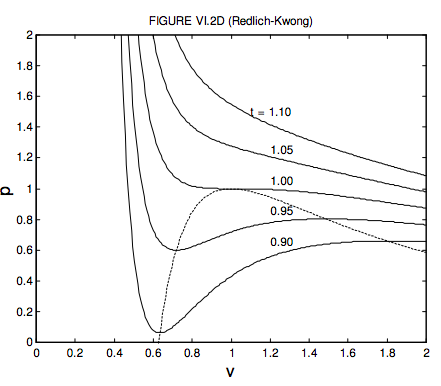
6.3: Van der Waals and Other Gases - Physics LibreTexts

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics
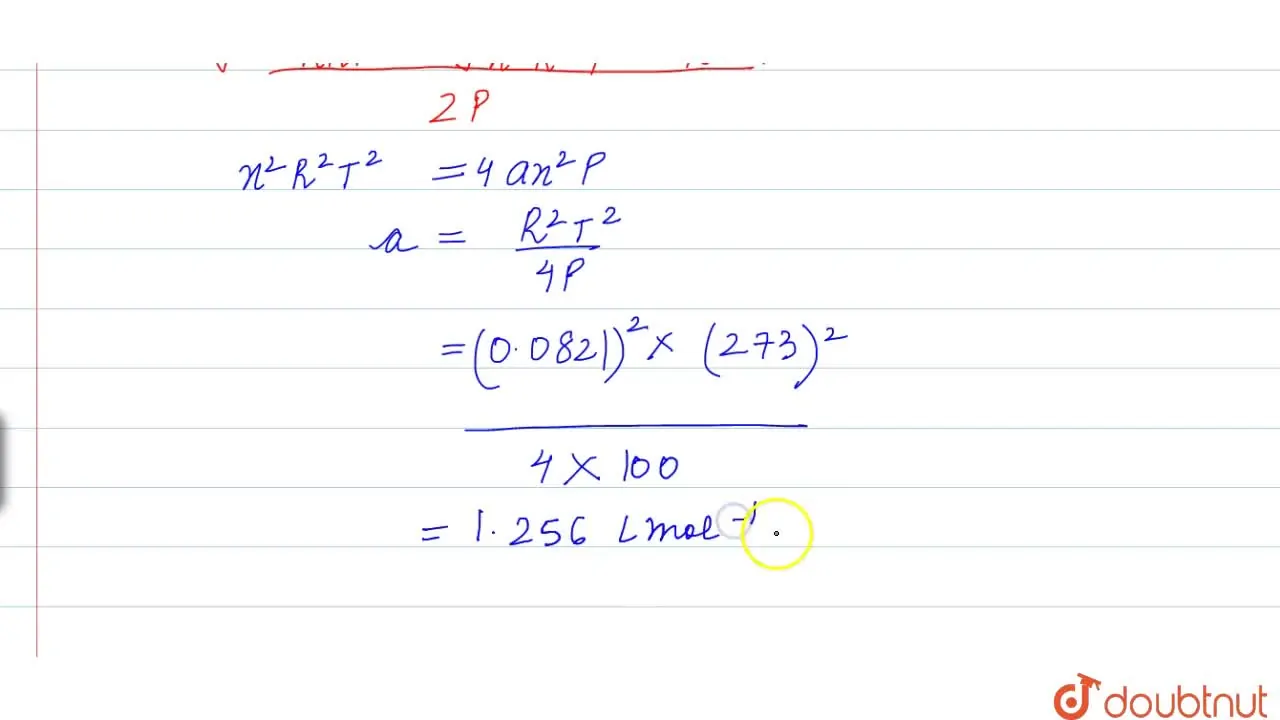
The compressibility factor for definite amount of van der Waals' gas a
How to calculate the values of critical pressure and temperature

The compression factor (compressibility factor) for 1 mol of a van der
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas

The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is

Solved 9 Compression factor Z Use the van-der-Waals equation

Bengali] What will the value of compressibility factor (Z) be for a g

Malayalam] The compressibility factor for definite amount of van der

Pick only the incorrect statement.for gas A, a=0,the compressibility factor is linearly dependent on pressure.for gas C,aneq 0,bneq 0,it can be used to calculate a and b by giving lowest P value.for

Solved 4.50 At low pressures the compressibility factor for

Compressibility factor (Z) for a van der Waals real gas at critical point is

The compressibility factor 1 mole of Vander Waal's gas 0^{o}C and 100 atm pressure is 0.447. Assuming the volume of gas molecules negligible, the value of Vander Waal's constant 'a' is: 1.24