The compressibility factor is Z = PV/R_g T. Evaluate

Answer to The compressibility factor is Z = PV/R_g T. Evaluate

Physical Chemistry The Compression Factor (Z) [w/1 example]

Compressibility factor - Wikipedia

Compressibility factor Z as function of temperature T with lines of

Real Gas - an overview

The compressibility factor of a gas is defined as Z=P V / R T. The compressibility factor of idea

Compressibility factor z versus 100/V, for several values of Pressure

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics

Guggenheim's Rule and the Enthalpy of Vaporization of Simple and Polar Fluids, Molten Salts, and Room Temperature Ionic Liquids
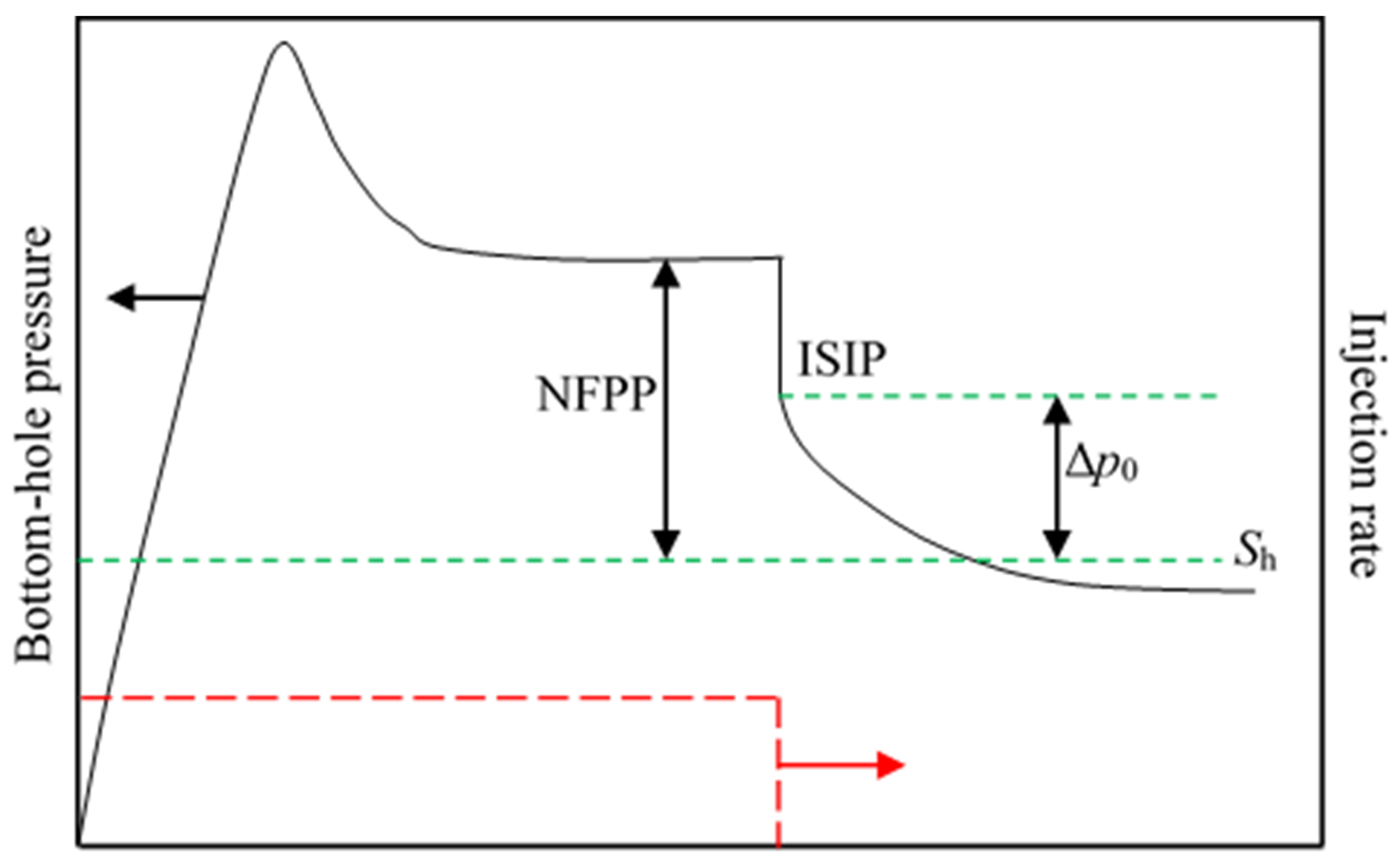
Processes, Free Full-Text