20.If Z is a compressibility factor, van der Waals equation at low
20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
20-If Z is a compressibility factor- van der Waals equation at low pressure can be written as

The van der Waals equation of state is given by (P +

Compressibility factor - Wikipedia

physical chemistry - Why do some gases have lower value of Z for a

At a high pressure, the compressibility factor (Z) of a real gas is us

Solved Non-Ideal gases (mixed methods) 1. Use the

gas laws - Graph of compressibility factor vs pressure when real


20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
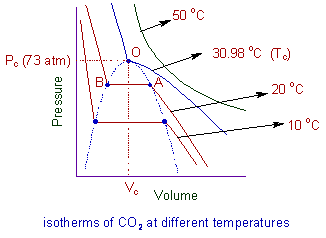
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR

If Z is a compressibility factor, van der Waals' equation at low press
The compressibility factor for one mol of a vanderwalls gas at 0